
The purpose of this site is to provide guidance and resources to ensure any research involving recombinant DNA or biological material is conducted in a manner that protects the health of researchers, the RIC community, and the environment.
- emaildbritt@ric.edu
Notice
Some of the guidance requires active participation of principal investigators to comply and adhere to the policies of RIC, Centers for Disease Control, National Institutes of Health, National Science Foundation, and Occupational Safety and Health Administration.
Program Details
Your compliance is crucial to the success of the goals of the RIC Biosafety Program. Those working with recombinant DNA (rDNA) and/or biological materials are required to submit a Biological Project Registration Form to the Institutional Biosafety Committee. In some cases the form must go through review before laboratory work may begin. The following activities are exempt from full review, but still require the submission of a Biological Project Registration Form.
rDNA that does not present a significant risk to health or the environment
rDNA that is not in organisms or viruses
rDNA consisting entirely of DNA segments from a single non-chromosomal or viral DNA source
rDNA consisting entirely of DNA from a prokaryotic host including its indigenous plasmids or viruses when propagated only in that host or when transferred to another host by well established physiological means.
rDNA consisting entirely of DNA from a eukaryotic host including its chloroplasts, mitochondria, or plasmids when propagated only in that host.
rDNA consisting entirely of DNA segments from different species that exchange DNA by known physiological processes.
The rDNA activities listed above are exempt from full review before labwork is initiated; however, a Biological Project Registration Form must be filed with the IBC.
Activities involving Biological Materials, work with live organisms, and animals:
Biosafety Level 1 (BSL1) materials are not known to cause disease in healthy adults are exempt from full review. A Biological Project Registration Form must be filed with the IBC.
Fluids, tissues, cells and other biological material of human or non-human primate origin are classified as BSL2. Projects involving BSL2 materials are not exempt from full IBC review and are not authorized until the protocols are reviewed and approved by the IBC.
Use of the following Select Agents in research must go through IBC approval before obtaining materials.
Abrin, Conotoxins, Diacetoxyscirpenol (DAS), Ricin, Saxitoxin, Tetrodotoxin, Shiga-like ribosome inactivating proteins, Botulinum neurotoxins, Clostridium perfringens epsilon toxin, Shigatoxin, Staphylococcal enterotoxins, T-2 toxin
Investigator Resources
Training is mandatory for faculty, staff, and students engaged in activities involving recombinant DNA and/or unfixed biological materials.
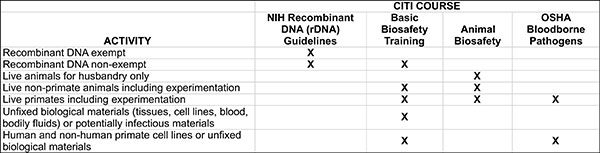
Training is through the Collaborative Institutional Training Initiative (CITI).
Instructions for training:
- Open a web browser and proceed to www.citiprogram.org
- Click the “New Users Register Here” link to create a new account.
- Section 1. Select your institution or organization: choose “Rhode Island College” from the Participating Institutions pull-down list. Leave the other pull-down lists blank.
- Section 2. Select your Username and Password: choose a username and password that you will remember and note them for your records.
- Section 3. Enter your name: put your first and last name.
- Section 4. Enter your email address: list a preferred email address.
- Click the Submit button at the bottom of the page to continue.
- Next page opens with a table entitled, “Please provide the following information requested by Rhode Island College.”
- Complete each row marked as mandatory with a red asterisk. Click the Submit button at the bottom of the page to continue.
- Next page is entitled, “Select Curriculum - Rhode Island College”. Here you enroll in one course for each learner group in which training is offered. Once your account is created you can enroll in as many courses as you want or need. There is no need to pick one course in each content area unless you have been specifically requested to do so. Click the Submit button at the bottom of the page to continue.
- Next page states “At this point you can register with another institution.” You can add affiliations at any point in the future. Most individuals should click the No button to remain affiliated with Rhode Island College only.
- Your account creation is complete. You may now take and enroll in courses, change learner groups and affiliations, and perform other account maintenance.
- Once you have completed a course the RIC Office of Sponsored Programs will receive confirmation.
The Institutional Biosafety Committee (IBC) has been established as specified in the NIH Guidelines for Research Involving Recombinant DNA Molecules and CDC Guidelines for Biosafety in Microbiological and Biomedical Laboratories. The RIC IBC oversees research involving recombinant DNA, and develops policies and provides expertise to reduce risks to faculty, staff and students working with biohazardous materials and recombinant DNA technology. RIC IBC reviews research protocols to determine whether a Principal Investigator (PI) who administers, handles or uses biological materials employs containment, decontamination, and disposal procedures in compliance with federal, state and local regulations, and ensures users are properly trained in good microbiological techniques.
IBC composition, standards, and procedures follow the NIH Guidelines For Research Involving recombinant DNA molecules Sections I-E-2 through IV-B-2-b-(9)). The RIC IBC is comprised of six members with expertise in recombinant DNA technology and the capability to assess any potential risk of research activities to public health or the environment; four members are RIC faculty and two members are not affiliated with RIC, as stipulated by the NIH Guidelines.
The RIC IBC provides resources, forms and templates at the IBC website (www.ric.edu/biosafety). Researchers can access this site to register their research involving recombinant DNA molecules and cell/tissue culture experiments. Once received, the IBC reviews research registration forms to ensure procedures are in place to protect faculty, staff, and students from biological materials.
RESPONSIBILITIES:
- Developing biosafety policies applicable to Rhode Island College activities, including work practices, biohazardous waste, and medical surveillance of personnel.
- Reviewing and approving proposed research in accordance with CDC/NIH guidelines.
- Setting required containment levels for research projects. Generally, the biosafety levels (BSLs) established by the CDC and NIH will be used as the level of containment; however, the IBC can increase or decrease the level of containment according to the specific circumstances of the project.
- Working with the Campus Police and the Facilities Department to recommend and review design specifications and criteria for existing and new containment facilities.
- Evaluate the public health and environmental risks associated with all biohazardous treatment and disposal methods, including disposal of these wastes according to provisions of the Rules and Regulations for Use of Wastewater Facilities Within the Narragansett Bay Commission District and Rhode Island Regulation DEM-DAH-MW-01-92.
- As necessary, make recommendations to management and changes to biohazardous waste management procedures.
Cell and Tissue Culture Biosafety
- Biosafety in Microbiological and Biomedical Laboratories (BMBL)
- American Biological Safety Association (ABSA) Risk Group Categories
- World Health Organization (WHO) Biosafety
- Health Canada Laboratory Biosafety Guidelines
- OSHA Bloodborne Pathogens
Recombinant DNA Safety
CDC/NIH Biosafety Levels
- BIOSAFETY 1 (BSL1) is suitable for work involving well-characterized agents not known to cause disease in healthy adult humans, and of minimal potential hazard to laboratory personnel and the environment.
- BIOSAFETY LEVEL 2 (BSL2) is similar to Level 1 and is suitable for work involving agents of moderate potential hazard to personnel and the environment.
- BIOSAFETY LEVEL 3 (BSL3) is applicable to clinical, diagnostic, teaching, research, or production facilities in which work is done with indigenous or exotic agents which may cause serious or potentially lethal disease as a result of exposure by the inhalation route.
- BIOSAFETY LEVEL 4 (BSL4) is required for work with dangerous and exotic agents which pose a high individual risk of aerosol-transmitted laboratory infections and life-threatening disease.
NIH Risk Group Levels
- Risk Group 1 (RG1) agents are not associated with disease in healthy adult humans.
- Risk Group 2 (RG2) agents are associated with human disease which is rarely serious and for which preventive or therapeutic interventions are often available.
- Risk Group 3 (RG3) agents are associated with serious or lethal human disease for which preventive or therapeutic interventions may be available.
- Risk Group 4 (RG4) agents are likely to cause serious or lethal human disease for which preventive or therapeutic interventions are not usually available.
